Potassium Citrate
1080mg modified release tablets
Established Efficacy, Better Compliance**
- First licensed potassium citrate for the treatment of recurrent kidney stones
- Absolute Risk Reduction of 50% for stone recurrence (6)
- Enables compliance with NICE Renal and Ureteric Stone guidelines (5)(NG118)
- Treatment cost per day £1.86*
*Mild hypocitraturia
**compared with currently available liquid and granule formulations.
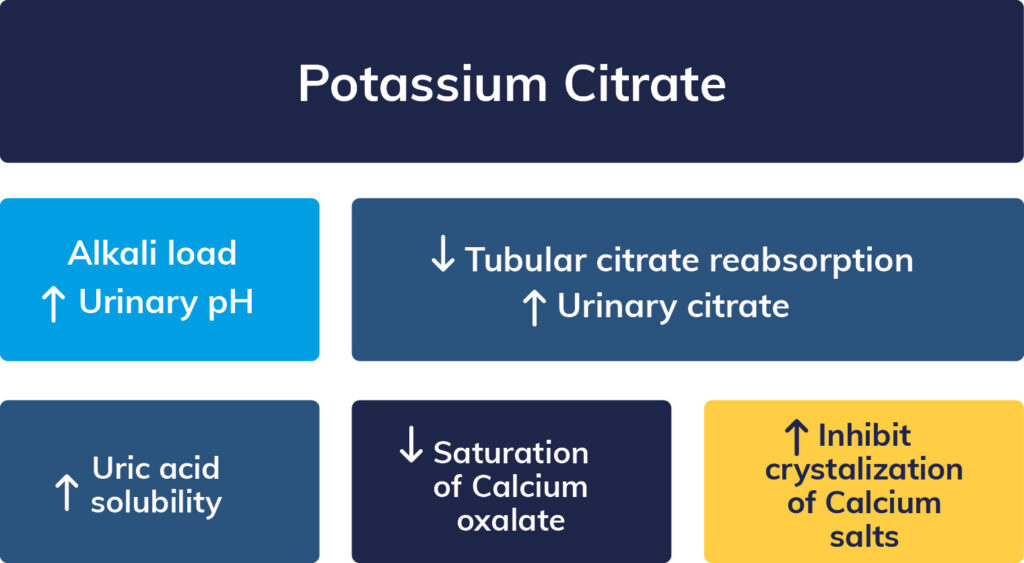
Mechanism of Action (7)
Potassium citrate inhibits the formation of kidney stones through the following mechanism:
Indicated for:
- The treatment of patients with kidney stones and hypocitraturia, or chronic calcium oxalate stones.
- The treatment and prevention of recurrent uric acid lithiasis with or without calcium lithiasis and cystine lithiasis.
- The treatment of renal tubular acidosis with calcium nephrolithiasis.
Take three times daily with meals.
Improved patient adherence via a palatable, convenient tablet formulation
Efficacy Data (6)
| Study | Year | Design | Population | Number of patients | Stone Type | Dose per day | Follow-up, years | ARR, % | RRR, % | Estimated NNT |
|---|---|---|---|---|---|---|---|---|---|---|
| Barceló et al. (1) | 1993 | RCT | Recurrent hypocitraturic adults | 57 | CaOx | 30–60 mEq | 3 | 52.2 | 65 | ≈2 |
| Ettinger et al. (2) | 1997 | RCT | Recurrent CaOx formers | 64 | CaOx | 42 mEq. potassium, 63 mEq. citrate | 3 | 50.7 | 79.7 | ≈2 |
| Soygür et al. (3) | 2002 | RCT | Post-SWL stone-free adults | 110 | CaOx | 60 mEq | 1 yr | 28.6 | 100 | ≈4 |
| Phillips et al. (4) | 2015 | Meta-analysis | Mixed recurrent stone formers | 477 | CaOx pre-dominant | Variable | 1-4 yrs | RRR ~75 | NA | ≈2-3 |
Table 1: Key studies of citrate therapy and estimated NNT by stone phenotype. ARR = Absolute Risk Reduction, RRR = Relative Risk Reduction; NNT = Number Needed to Treat, SWL = extracorporeal Shock Wave Lithotripsy, CaOx= Calcium Oxalate
Guidelines
Renal and Ureteric Stone guidelines from NICE recommend use of potassium citrate alongside diet and lifestyle advice for prevention of stones in patients with predominantly calcium oxalate stones. (NICE, NG118). Until now the recommendation has been based on the off-label use of products that are indicated for other conditions.

Prescribing Information Renodyra 1080 mg modified-release tablets
For the treatment of adult patients with kidney stones and hypocitraturia or chronic calcium oxalate stones, prevention and treatment of recurrent uric acid lithiasis with or without calcium lithiasis and cysteine lithiasis and treatment of renal tubular acidosis with calcium nephrolithiasis. The tablets should be taken orally with meals or within 30 minutes after meals up to three times a day in divided doses. Dosing is based upon severity and starts from 3240mg per day up to 10800mg maximum dose. Do not use: If hypersensitivity to the active substance or to any of the excipients, renal impairment, active or persistent urinary tract infections, significant or complete obstruction of the urinary tract, hyperkalaemia, severe myocardial injury, uncontrolled diabetes mellitus, adrenal insufficiency, metabolic or respiratory alkalosis, active peptic ulcer, delayed gastric emptying or intestinal obstruction. This medicine should be used with caution when combined with other products that increase plasma potassium or predispose to cardiac arrest and regular monitoring of renal function parameters and blood potassium levels is recommended every 4 months. In patients with severe renal function, regular monitoring at least twice a year is required. Common side effects include abdominal pain, nausea, diarrhoea, gaseousness, dyspepsia, oesophagitis, dysphagia and skin rash. Serious adverse reaction such as hyperkalaemia may lead to muscle weakness/paralysis, cardiac conduction abnormalities and cardiac arrhythmia. Prescribers should consult the summary of product characteristics for further information. The product is packed in a polyethylene (HDPE) bottle, closed with a tamper evident cap with an aluminium PE / PP / Al insert. Legal category: POM PL56328/0002 MAH: Penlan Pharmaceuticals Ltd, 45-47 Monument Hill, Weybridge, KT13 8RN Price: £62.04 Revised: December 2025
UK/100036/013 v2.0 December 2025
References:
- Barcelo, P., Wuhl, O., Servitge, E., Rousaud, A., & Pak, C. Y. C. (1993). Randomized Double-Blind Study of Potassium Citrate in Idiopathic Hypocitraturic Calcium Nephrolithiasis. Journal of Urology, 150(6), 1761–1764. https://doi.org/10.1016/S0022-5347(17)35888-3
- Ettinger, B., Pak, C. Y. C., Citron, J. T., Thomas, C., Adams-Huet, B., & Vangessel, A. (1997). Potassium-Magnesium citrate is an effective prophylaxis against recurrent calcium oxalate nephrolithiasis. Journal of Urology, 158(6), 2069–2073. https://doi.org/10.1016/S0022-5347(01)68155-2
- Soygür, T., Akbay, A., & Küpeli, S. (2002). Effect of Potassium Citrate Therapy on Stone Recurrence and Residual Fragments after Shockwave Lithotripsy in Lower Caliceal Calcium Oxalate Urolithiasis: A Randomized Controlled Trial. Journal of Endourology, 16(3), 149–152. https://doi.org/10.1089/089277902753716098
- Phillips, R., Hanchanale, V. S., Myatt, A., Somani, B., Nabi, G., & Biyani, C. S. (2015). Citrate salts for preventing and treating calcium containing kidney stones in adults. Cochrane Database of Systematic Reviews, 2015(10). https://doi.org/10.1002/14651858.CD010057.pub2
- NICE Renal and ureteric stones: assessment and management (NG118), January 2019
- Data on File 100036 v1.0 January 2026
- Youssef, R., Preminger, G., Lipkin, M. (2014). Potassium Citrate and Calcium Stones: Benefit or Risk?. In: Pearle, M., Nakada, S. (eds) Practical Controversies in Medical Management of Stone Disease. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-9575-8_9
Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk. Adverse events should also be reported to Penlan Pharmaceuticals Ltd.

